Fission reaction equation1/7/2024 
Pössel goes on mentioning that the strength of the nuclear bond depends on the number of neutrons and protons involved in the reaction. “The main contribution is due to binding energy being converted to other forms of energy – a consequence not of Einstein’s formula, but of the fact that nuclear forces are comparatively strong, and that certain lighter nuclei are much more strongly bound than certain more massive nuclei.” Associated with all of these forces are what is called binding energies – the energies you need to supply to pry apart an assemblage of protons and neutrons, or to overcome the electric repulsion between two protons.” Nuclear binding energy curve. Then, there are further forces, for instance the electric force with which all the protons repel each other due to the fact they all carry the same electric charge. 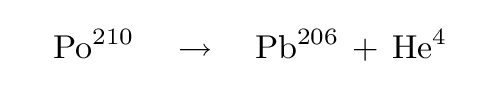
First of all, there are the nuclear forces binding protons and neutrons together. In order to understand nuclear fission (or fusion), it is necessary to examine the bonds between these components. They have component parts, namely protons and neutrons. Atomic nuclei aren’t elementary and indivisible. “To see where the difference lies, one must take a closer look. Markus Pössel, the managing scientist of the Center for Astronomy Education and Outreach at the Max Planck Institute for Astronomy in Heidelberg, Germany, provides us with a great explanation for why nuclear reactions can be violent. It can only follow that the square of the speed of light only partly explains the huge difference in energy released between nuclear and chemical reactions. We’ve all burned a match and there was no mushroom cloud, though. Multiply it by c 2 and you’ll wind up with the energy set free during the chemical reaction. The same formula applies, so if you measure the difference between the rest mass of unburned material and the rest mass the burned object and gaseous byproducts, you’ll also get a tiny mass difference. A common example is spontaneous oxidation or, more familiarly, combustion. Of course, this conservation of energy holds true across all domains, both in relativistic and classical physics. This mass discrepancy might be tiny but once you multiply it by c 2 (the speed of light squared), the equivalent energy can be huge. This difference is called the ‘mass defect’ and it’s precisely this missing matter that’s been converted into energy, the exact amount computable using Einstein’s famous equation. Interestingly, when you count all the particles before and after the process, you’ll find the total mass of the latter is slightly smaller than the former. By the process through which fission splits uranium atoms, for instance, a huge amount of energy, along with neutrons, is released. It didn’t take scientists too long to realize there was a massive amount of energy waiting to be exploited. We now had a new way to measure a system’s total energy simply by looking at mass, which is a super-concentrated form of energy. Millions of people recognize it and can write it down by heart even though they might not understand anything about the physics involved.īefore Einstein, mass was considered a mere material property that described how much resistance the object opposes to movement. For Einstein, however, relativistic mass - which now takes into account the fact that mass increases with speed - and energy are simply two different names for one and the same physical quantity. It’s been featured in countless novels and movies. It’s printed on coffee mugs and T-shirts. This deceivingly simple equation can be found everywhere, even in pop culture. On November 21, 1905, physicist Albert Einstein published a paper in Annalen der Physik called “ Does the Inertia of a Body Depend Upon Its Energy Content?“ This was one of Einstein’s four Annus Mirabilis papers (from Latin, annus mīrābilis, “Extraordinary Year”) in which he described what has become the most famous physical equation: E = mc 2 (energy equals mass times the velocity of light squared). 
ADVERTISEMENT From Einstein to nuclear weapons
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
